 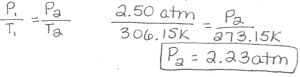
A look at the nutrition facts show common processed corn products, like corn oil and high fructose corn syrup, lose most of the beneficial fiber and nutrients during processing. The vast majority of that corn is not the hard to digest kernels nibbled off the cob, but corn that's been turned into soft tortillas, chips, popcorn and - the big one - high fructose corn syrup.Įasier to digest, however, is not to be confused with healthier. The Tufts University report estimates that each American consumes 160 lbs. In fact, the majority of the corn you eat is processed. Grinding, wet milling, cooking - every processing step breaks down those hard to digest fiber molecules a little further, she said. That's true for humans and animals alike. "The more you process it, the easier it is to digest," Watson said. There's a way to make corn more digestible and disappear from your poop altogether: processing. What's the difference between a fruit and a vegetable? Why do some fruits and vegetables conduct electricity? a and b e.- Why are bananas berries, but strawberries aren't? The water displacement method used determines the volume of a. Assuming all the water has evaporated, why do you still need to let your flask cool before weighing? 7. therefore, the ultimate calculation of pressure if you did not make sure all the water was evaporated from inside the flask before weighing is: a. the calculated temperature of water in the kernels will be: a. the calculated moles of water in the kernels will be: a. If you did not make sure all the water was evaporated from inside the flask before weighing: A the calculated volume of water in the kernels will be: a. therefore, the ultimate calculation of pressure if not all the popcorn kemels popped is: a. 
the calculated volume of water in the kemels will be: a. If not all the popcorn kernels popped, A. If we combine each of these individual relationships, we come up with the Ideal Gas Law which allows us to relate all four variables to each other. Volume is directly proportional to the number of moles of gas as well as the temperature on the Absolute (Kelvin) scale and inversely proportional to the pressure of the gas. The Ideal Gas Law is derived using the individual relationships between volume and the other variables. These variables are related to cach other and the Ideal Gas Constant (R) (Equation 1). PV = nRT (Equation 1) Gases are fully described by four variables: pressure (P), volume (V), number of moles (n), and temperature (T). The popcorn then "explodes" releasing the starchy substance as the white fluffy part of the com The outer sha Each wel holde 3 min order to po The starch The Leroes POPS going through the vapenation process The shell builds up pressure and The shell begin to What pressure is required to pop the corn? In this lab, we will determine the pressure required to pop com using our understanding of the Ideal Gas Law (Equation 1). When the gas expands enough to fill its container (the hull), the pressure increases until great enough to overcome the rigidity of the outer shell. The liquid water turns to steam as it is heated and, according to Charles law, continues to expand as the temperature increases. Transcribed image text: Lab 5: The Pressure of Popping Corn Why does popcom pop? The outer shell or hull of a kernel of popping com surrounds a drop of moisture and a starchy substance. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
